OUR PIPELINE
Developing next-generation polyene therapy

EL219 for IV infusion is a novel, next-generation polyene. In nonclinical and Phase 1 human studies, EL219 has demonstrated an improved safety profile. Potent in vitro activity and in vivo efficacy is observed against a broad range of fungi, including Aspergillus species with reduced susceptibility to polyenes.
FDA has granted Qualified Infectious Disease Product (QIDP) and Fast Track designation to EL219 for
- Empiric antifungal therapy for presumed invasive fungal disease
- Treatment of invasive aspergillosis
- Treatment of cryptococcosis
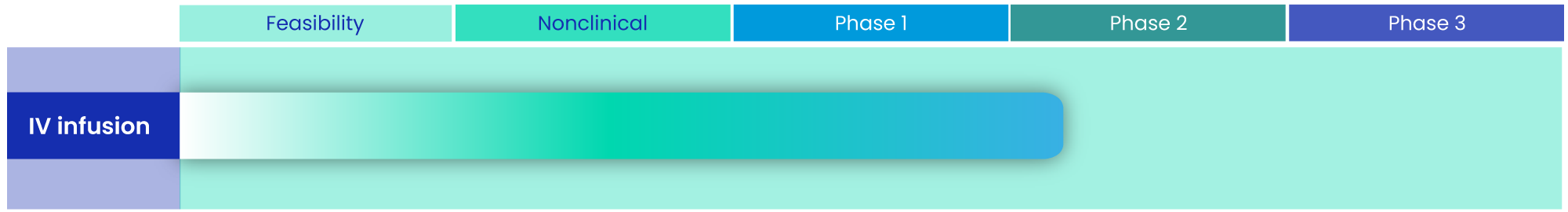
Our EL219 clinical program
Qualified Infectious Disease Product Designation: A US regulatory status granted by the FDA to certain antibacterial and antifungal drug candidates intended to treat serious or life-threatening infections. Established under the Generating Antibiotic Incentives Now (GAIN) Act in 2012 to incentivize the development of new drugs to combat antibiotic resistance, the designation offers the potential for expedited development and regulatory review.
Fast Track Designation: An FDA program designed to expedite the development and review of new drugs and biologics that address unmet medical needs for serious and life-threatening conditions.